I am vaccinated.
I chose the 'tried & true' type vaccine rather than a new formula (mRNA)
I have more faith in the older type vaccine.
In defense of the anti-vax crowd,
We need to remember that thses mRNA types are NOT fully approved - emergency use only.
The studies are still ongoing for a final, full approval.
Today's NYTimes has a good article on this - for those interested
Most important is that you have protection to avoid the worst, hospitalization, intubation death.
1. mRNA vaccine technology is not entirely new
Vaccines such as the inactivated polio vaccine, or most flu vaccines, use inactivated viruses to trigger a person's
immune system to respond to that disease-causing organism. In other vaccines, such as the hepatitis B vaccine, an individual protein made by that organism is injected instead to trigger a similar response.
mRNA vaccines, however, trick the body into making the viral protein itself which, in turn, triggers an
immune response.
Although the COVID-19 vaccines made by Pfizer/BioNTech are the first mRNA vaccines to complete all clinical trial stages and be licensed for use, the technology has been around for a while.
Human trials of cancer vaccines using the same mRNA technology have been taking place since 2011. "If there was a real problem with the technology, we'd have seen it before now for sure," said Prof. Goldman.
Because the technology can be deployed extremely rapidly, and clinical trials have been so successful, mRNA platforms will be an important means of preparing for future epidemics, he says.
2. mRNA vaccines do not alter your DNA
A concern that some have had about the mRNA vaccines is that they could change people's DNA. But that idea is 'completely false' and has 'no scientific basis," says Prof. Goldman.
"The (vaccine) mRNA will not enter the nucleus of the cells, where our DNA is."
Once the injected mRNA enters a human cell, it degrades quickly and only stays in the body for a couple of days. This is why people need two injections to develop the best immune response, he says.
3. mRNA vaccines are very specific
The novel
coronavirus, or SARS-CoV-2, has a complex structure, and different parts of the virus trigger the immune system to produce different antibodies to neutralise the virus.
If an unvaccinated person catches the virus, they will produce antibodies that prevent the virus from entering human cells. They may also generate antibodies that do not have much impact. And in some cases, a person may produce antibodies which actually help the virus enter cells.
mRNA vaccines are much more specific. They are designed to only trigger an immune response to the virus's spike protein, which is just one component of the viral membrane and enables the virus to invade our cells.
To be sure this is the case, researchers are carefully monitoring that the vaccine does not trigger an unwanted immune response.
"So far this has not been shown for the (COVID-19) vaccines." But it 'will remain important to ensure the immune response triggered by the vaccine is focused on the viral spike protein," said Prof. Goldman.
The world's first mRNA vaccine has begun its rollout after being produced at unprecedented speed as part of the global effort to end the COVID-19 pandemic. A second one is hot on its heels. The two—one made by Pfizer/BioNTech and the other by Moderna—mark the first time this vaccine technology...

medicalxpress.com
History
Early research
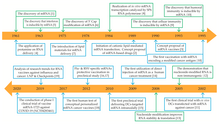
Timeline of some key discoveries and advances in the development of mRNA-based drug technology.
In 1989, the first successful transfection of mRNA packaged within a liposomal nanoparticle into a cell was published.
[3][15][16] In 1990, "naked" (or unprotected) mRNA was injected into the muscle of mice.
[3][17] These studies were the first evidence that
in vitro transcribed mRNA could deliver the genetic information to produce proteins within living cell tissue.
[3] At this time mRNA was proposed for immunization purposes.
[18][19]
In 1993, liposome-encapsulated RNA was shown to stimulate
T-cells in vivo, and in 1994, RNA proved useful as a vaccine to elicit both humoral and cellular immune response against a pathogen.
[3][20][21]
Development
In 2005, successful application of
modified nucleosides as a medium to get mRNA inside cells without setting off the body's defense system was reported.
[3][22] In 2010, the mRNA-focused biotechnology company,
Moderna, was started to develop mRNA biotechnologies.
[22][3]
In 2010, US government agency
DARPA launched a biotech research program called ADEPT as part of its mission to develop emerging technologies for the
US military.
[23] In 2011, DARPA recognized the potential of nucleic acid technology for defense against pandemics and began to invest in the field through ADEPT.
[23][24] DARPA's grants were seen as a vote of confidence which in turn encouraged other government agencies and private investors to also invest in mRNA technology.
[24] In 2013, DARPA awarded a $25 million grant to Moderna.
[25]
mRNA drugs for cardiovascular, metabolic and renal diseases, and selected targets for cancer were initially linked to serious side effects.
[26][27] mRNA vaccines for human use have been studied for
rabies,
Zika virus disease,
cytomegalovirus, and
influenza.
[28]
Acceleration
In December 2020, Moderna and BioNTech obtained FDA
emergency use authorization for their mRNA-based
COVID-19 vaccines, which had been funded by
Operation Warp Speed.
[22] On 2 December 2020, seven days after its final eight-week trial, the UK's
Medicines and Healthcare products Regulatory Agency (MHRA), became the first global medicines regulator
in history to approve an mRNA vaccine, granting emergency authorization for Pfizer–BioNTech's BNT162b2 COVID-19 vaccine for widespread use.
[7][8][29] On 11 December 2020, the
FDA gave emergency use authorization for the Pfizer–BioNTech COVID-19 vaccine.
[30]

en.wikipedia.org